Predators and Puppeteers

BIOS-SCOPE scientist Ben Temperton loads a DNA sample into the MinION for sequencing. Image courtesy of Ben Temperton
Scientists estimate there are more than a million times more viruses in the ocean than stars in the universe. While wildly abundant, their tiny sizes present a big a hurdle to fully understanding their function in ocean ecosystems. If a cell were the size of a baseball stadium, a virus would be roughly the size of a baseball, so not only are viruses difficult to see under the microscope, but even gathering enough of their genetic material to analyze can be tricky.
And because viruses pack a lot of genetic information into such a small space, deciphering their genetic complexity presents unique computational challenges as well.
This past summer, BIOS-SCOPE (Bermuda Institute of Ocean Sciences – Simons Collaboration on Ocean Processes and Ecology) scientists from the University of Exeter in the United Kingdom pioneered the use of a sleek new genetic sequencing device to discover which viruses impact microbial communities and carbon cycling in the Sargasso Sea. The instrument, an Oxford Nanopore Technology’s “MinION,” is the size of stapler, runs off a laptop USB plug, and offers an entirely new way to sequence DNA in the field.
DNA stores the instructions for life in a code made up of four chemical bases. The unique sequences created from these four bases give rise to the diversity of life on Earth, and DNA sequencing has already revealed a world of viruses and microbes that were once invisible to humans.
The MinION works by pulling a strand of DNA through a nanoscopic pore, which is just wide enough for large molecules to pass through. As the DNA moves through the pore, it produces a measureable electrical signal.

To extract enough viral DNA for sequencing, University of Exeter graduate student Joanna Warwick-Dugdale concentrates the viruses from 40 liters of seawater onto a filter. Image courtesy of Ben Temperton
“What you get in real time is called ‘squiggle data,’ which represents how the current is disturbed by each base of the DNA,” said University of Exeter graduate student Joanna Warwick-Dugdale, who brought the MinION to BIOS this spring and collected and analyzed virus samples from the BIOS-SCOPE cruise this summer.
It requires a lot of computational power to translate that squiggle into what bases went through the pore and reconstruct the entire DNA sequence. Consequently, Warwick-Dugdale has been concentrating viruses in her samples and extracting their DNA while at sea, but checking the quantity and quality of the DNA back on shore at BIOS before sequencing with the MinION.
“With the MinION, the quality of DNA is incredibly important. Anything where the DNA is wonky, such as a slightly broken strand, won’t go through the pore,” said BIOS-SCOPE bioinformatician Ben Temperton, who is also Warwick-Dugdale’s advisor at the University of Exeter. Though somewhat finicky, the MinION can read chunks of DNA that are 100,000 base-pairs long, whereas high-throughput sequencing (also known as “next generation” sequencing, due to its speedy and relatively cheap ability to generate billions of reads) requires chunks of DNA that are less than 600 base-pairs long.
“With next generation sequencing, it’s like taking a whole library of books and shredding them into 3-5 word chunks, then trying to pull the scraps of paper together and realign them into their original form,” said Temperton. “We’re getting pretty good at reassembling cellular genomes this way, but with viral genomes, sometimes they can’t be reassembled at all. In comparison to next-generation sequencing, assembling MinION data is like trying to recreate the books from whole pages.”
Critically, the longer MinION reads span regions of high variability, which are common in viral genomes. In contrast, the short reads generated by next generation sequencing can get so scrambled that they are impossible to put back together.
“By using these long reads, we are getting better at identifying what’s out there, and it’s a big step up in terms of how accurately we’re representing the diversity of viruses,” Temperton said.
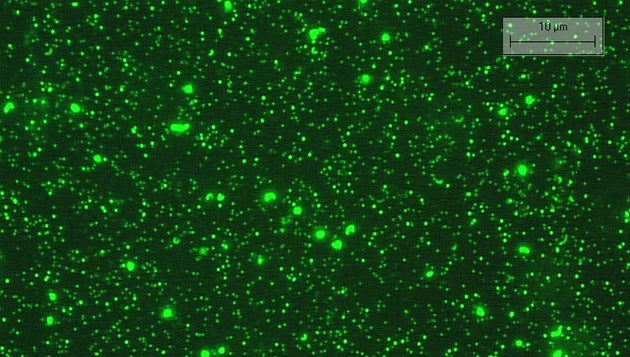
The multitude of viruses in seawater can be counted once they are magnified 1,000 times under the microscope, and treated with a fluorescent stain that attaches to their DNA. Image courtesy of Rachel Parsons
Tiny predators, big impact
As predators of marine bacteria, viruses (so-called bacteriophages; literally, “bacteria eaters”) are powerful players in nutrient cycles. Every day, viruses infect 10 to 20 percent of all the bacteria in the ocean, hijacking bacterial cellular processes to produce more viruses. Once an infected bacterium is brimming with the next generation of viral particles, it is programmed to self-destruct. New viral particles spill into the seawater in a soup of dissolved organic matter, and the carbon and nutrients that were contained by the bacteria—and once destined to move up the food chain, should that bacteria have been eaten—suddenly become sustenance for other microbes. Thus, viral predation may actually fuel the microbial communities at the base of the ocean’s food web.
But viruses are also crafty. When bacterial populations collapse, there is little benefit to killing their remaining hosts. Some viruses adjust their infection strategy by integrating their DNA into the DNA of their host bacteria. In this way, they can become puppeteers, changing the day-to-day activities of the bacteria in ways that benefit the virus, but can also change the bacteria’s role in the ecosystem. For example, some viruses carry genes for photosynthesis and carbon metabolism; by integrating into their hosts, they may be influencing global biogeochemical cycles at key times.
While BIOS-SCOPE microbiologist Steve Giovannoni of Oregon State University sequences entire genomes from single cells to better understand the ocean’s most abundant bacteria, Temperton can also comb through these bacterial sequences to hunt for viruses. By matching the viral sequences embedded in bacterial genomes with viral sequences recovered from the ocean, the team can begin to identify which parts of the bacterial community are targeted by specific viruses.

To study the microbial community inhabiting seawater samples, Rachel Parsons prepares reagents for a protocol that uses fluorescent probes to label unique microbial groups, so that they can be identified and counted under the epifluorescent microscope. Photo by Michael Parsons
Following clues from a decade of BATS data
Some of the seminal insights into the ocean’s viral communities have come from work at BIOS. In 2012, BIOS microbial research specialist Rachel Parsons, University of California-Santa Barbara microbial oceanographer (and BIOS-SCOPE Program Director) Craig Carlson, and their colleagues published a study of viruses at the Bermuda Atlantic Time-series Study (BATS) that revealed regular seasonal patterns in marine virus populations.
“It was a huge undertaking initially to get viral abundance for our samples,” Parsons explained. Glowing like millions of stars in the night sky, viruses can be enumerated under the microscope using a fluorescent stain that attaches to the minuscule amount of DNA encapsulated within each virus. However, Parsons said the sheer abundance of viruses made it “absolutely mind-numbing” to count viral particles one by one. She worked to establish an automated protocol, whereby fluorescent viruses imaged under the microscope could by analyzed by a computer.
“Once that was up and running, I loved viruses,” Parsons said. She ended up collecting an unprecedented decade’s worth of viral abundance data in BATS samples, on top of her standard surveys of marine bacteria (also collected via microscopy, using fluorescent markers to identify bacteria of interest).
The BIOS team had expected that viral abundance would mirror the abundance of the bacteria in the water—after all, predators require prey. However, they found that maximum viral abundance occurred each fall at roughly 250 to 325 feet (80 to 100 meters) below the surface, which didn’t mirror populations of the dominant bacteria SAR11 or other major bacterial groups they were tracking.
An “ah-ha!” moment came when BIOS colleague Mike Lomas (now at Bigelow Laboratory for Ocean Sciences) gave a talk presenting the annual distribution of an abundant, photosynthesizing cyanobacteria called Prochlorococcus at the BATS station. The patterns in Prochlorococcus abundance visually matched the patterns of viral abundance—peaks at the same depth, during the same seasons—and statistical analyses showed a strong relationship between the two over time, suggesting the most abundant viruses at the BATS station may be targeting this tiny microbe at the base of the food web.
“With this huge dataset we got an idea of what viruses might be doing, but we couldn’t prove exactly which viruses were there,” said Parsons. “Now that molecular tools have developed further, we can return to this question with BIOS-SCOPE.”
Tackling the microbial web, from top to bottom
The microbial community gets “chewed down” each day by a combination of predation from larger organisms and predation from viruses. Predation is considered the dominant ‘top-down’ control on ecosystem processes, while the availability of nutrients controls ecosystem processes from the ‘bottom-up.’ Of course, the activities of these predators—viruses or otherwise—spill nutrients into the water, meaning they can influence communities from the ‘bottom-up’ as well.
With marine chemists and microbiologists on the BIOS-SCOPE team, the nutrients and organic matter being consumed and produced are rigorously characterized on BIOS-SCOPE cruises. And complementing the Temperton lab’s studies of viruses, BIOS-SCOPE visiting investigator Elizabeth Harvey, from the University of Georgia’s Skidaway Institute of Oceanography, has been working on ship-board experiments to study how quickly single-celled microbial predators consume bacteria and phytoplankton. Moving up the food chain, the ecosystem’s zooplankton predators are being studied by BIOS zooplankton ecologists Leocadio Blanco-Bercial and Amy Maas.
“The strength of BIOS-SCOPE is that this fantastic team is really getting a holistic view of how the ecosystem works,” Temperton said.
